Biotic Antibiotics – Phages to the Rescue ?
 Thousands of hospitals and communities are now grappling with the fruits of our success with antibiotics… deadly bacterial resistance. Some people think methicillin resistant staph aureus (MRSA) and its terrifying descendant vancomycin resistant staph aureus (VRSA) are threatening to return the world to the dark ages before penicillin and sulfa were discovered.
Thousands of hospitals and communities are now grappling with the fruits of our success with antibiotics… deadly bacterial resistance. Some people think methicillin resistant staph aureus (MRSA) and its terrifying descendant vancomycin resistant staph aureus (VRSA) are threatening to return the world to the dark ages before penicillin and sulfa were discovered.
Now, some researchers are looking back in time to the early 20th century to find treatments that can potentially overcome the problems of bacterial antibiotic resistance and can give us new paradigms in the treatment of infectious diseases. The idea of using viruses as therapeutic agents may even go beyond treatment of bacterial infections to the potential of treating other important and debilitating illnesses such as Alzheimer’s disease.
Actually, antibiotics have been around in some form for millenia. In 3500 BC the Sumerian doctors gave sick patients beer soup mixed with snakeskins and turtle shells, doctors in Babylon healed eye infections with an ointment made of frog bile and sour milk and the Egyptians used honey and molds to treat infected wounds. The ancients thought such curative effects were ‘magic’.
In the 19th century, Pasteur was able to demonstrate that many illnesses were actually caused by microbes. Then, in 1928 Alexander Fleming observed bacterial inhibition by a mold on a neglected petri dish and launched the modern antibiotic era.
The success of antibacterial antibiotics ( from penicillin to the quinolones ) over the past 80 years is unquestioned, but the current antibiotic era may be coming to an end. Increasingly, the rapid spread of antibiotic resistance and the evolution of ‘superbugs’ has made researchers look back in time to other approaches to the treatment of infection. Harnessing viruses that attack harmful bacteria may be the next phase of the struggle to control infection in humans. Research presented at the Bacterophage 2008 Meeting in Hertfordshire, UK confirmed the effectiveness of using antibacterial viruses in the treatment of chronic Pseudomonas ear infections that were resistant to conventional antibiotic treatment.
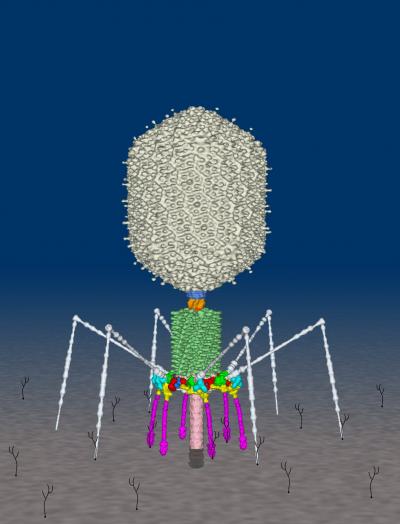
Bacteriophages, viruses which attack bacteria, were discovered in 1916 by Félix d’Herelle, a bacteriologist working at the Pasteur Institute in Paris. He noticed that soldiers suffering from bacterial dysentery had an enigmatic microbe which always appeared in the stools of patients just before they began to recover from the disease. He named these mysterious microbes bacteriophages (literally ‘bacterial eating’), and noted that they dissolved even highly concentrated cultures of the dysentery bacteria (Shigella) on his culture plates. D’Herelle concluded that these phages had something to do with the healing process of bacillary dysentery and he tried to harness their power for fighting infections. (See a video showing a T4 Bacteriophage attacking a bacteria by clicking here ).
Felix d’Herelle treated chickens infected with fowl typhoid after he discovered a specific phage virus against Salmonella gallinarum ( the causative agent of fowl typhoid). He then used phage therapy to treat four children in Paris suffering from severe cases of bacterial dysentery in 1919. All the children recovered (although it was never clear whether the recovery was related to the introduction of the bacteriophage viruses as it was not a controlled experiment).
For the next two decades, phage therapy was used in many countries to treat millions of patients against infectious diseases like cholera, bacillary dysentery and boils. But after penicillin was widely introduced in the 1940s, phage therapy lost favor and was largely forgotten by western medicine.
However, the treatment did survive in pockets in Eastern bloc countries alongside the use of antibiotics. Research continued in the Soviet Union and in outlying regions like the present Republic of Georgia. Now, since the fall of the Soviet Union and the increasing concern about antibiotic resistance, western medicine is beginning to take an interest in its potential. One company specializing in phage therapy is the Phage Therapy Center in the Republic of Georgia. ( Watch a captivating BBC video about phage therapy in the Republic of Georgia here ).
 Phage therapy has several potential advantages over antibiotics. One of them is that phages are very specific to the bacteria they invade and do not harm the useful bacteria that live in and on the body. Most of the antibiotics we currently use also attack harmless and even beneficial bacteria that make a living on us. This can lead to severe diarrheal infections or colonization by other undesirable organisms (such as the current hospital spread of Clostridium difficile), especially in patients with a weak immune system.
Phage therapy has several potential advantages over antibiotics. One of them is that phages are very specific to the bacteria they invade and do not harm the useful bacteria that live in and on the body. Most of the antibiotics we currently use also attack harmless and even beneficial bacteria that make a living on us. This can lead to severe diarrheal infections or colonization by other undesirable organisms (such as the current hospital spread of Clostridium difficile), especially in patients with a weak immune system.
Phages are quite specific to the particular bacteria they search out and destroy. Furthermore, phages can attack bacteria ( such as MRSA, Clostridium difficile and Listeria monocytogenes ) that show resistance to conventional antibiotics. Phages only attack bacteria and have absolutely no other apparent effects on human, animal, insect, or plant cells. Even with bacteria, a phage kills only the strains or a subgroup of strains within the targeted bacterial species, which makes very specific therapy possible.
Phages are not genetically engineered or altered in any way, but rather, are found in nature in incredible abundance. Phages are probably the most widely distributed biological entity in the biosphere, with an estimated population of >1030 or ~10 million per cubic centimeter of any environmental niche where bacteria reside. This provides a huge, naturally available pool of potentially useful bacteriophages.
Western medicine is once again beginning to investigate the possibilities of this mode of therapy. At a recent Society for General Microbiology meeting in Edinburgh, researchers presented work on incorporating bacteriophages into dressings for wounds and cleaning materials used in hospitals. The scientists found a way to bind the phages to polymer particles, allowing the viruses to remain active for up to three weeks rather than breaking down after a few hours. Use of phages has been reported for the treatment of post-surgical infections. In August, 2006 the United States Food and Drug Administration (FDA) approved using bacteriophages on cheese to kill the Listeria monocytogenes bacteria, giving them GRAS status (Generally Recognized As Safe). In July 2007, the same bacteriophages were approved for use on all food products.
Yet, there are potential problems with phage therapy. In order to use phages for treatment, you have to know precisely which bacteria are causing the infection. As those of us in the medical community know, that is not always apparent, at least initially. Thus, when the patient is often the sickest and most in need of treatment, ‘shotgun dosing’ with a number of phages aimed at the most likely pathogens might be necessary. Inactivation of many phages by gastric acid could limit the use of oral tablets to administer phage therapy. Also, there is always the problem of natural selection. As with all types of treatments against living, mutating organisms, resistance can develop to bacteriophage therapy just as it can to conventional antibiotic therapy. But advocates feel these drawbacks are minor, and do not present major stumbling blocks to expanding the use of phages for medical treatment.
Some researchers suggest that by selecting only phages that lyse (actually kill, rather than just inactivate) the bacteria, the development of resistance can be slowed. Also, introducing ‘cocktails’ containing multiple phages directed at a single bacterial species ( like combining multiple antibiotics for drug resistant strains of Tb) can lessen the chances of rapid resistance developing. One group of plant biology researchers have demonstrated the success of this approach using phage therapy to combat ‘Bacterial Blight’ in geraniums.
Recently, phages have been investigated in the treatment of other disease processes. An Israeli scientist, Beka Solomon has demonstrated that bacteriophages can dissolve plaques that develop in the brains of mice (similar to the plaques that develop in humans with Alzheimer’s disease). Solomon first began thinking about sending phages through the nasal passages because the plaque that causes Alzheimer’s first appears in the olfactory bulb. As a result, one of the early symptoms of this devastating disease is often loss of smell. She administered non-lytic filamentous phages in small doses through the mouse nasal passages, thus giving the phages a direct and rapid route to the brain (circumventing the ‘blood-brain barrier’ which prevents many treatments from even getting into the brain). She found certain phage viruses can lock onto the extracellular amyloid plaques associated with Alzheimer’s and dissolve them, reducing inflammation in the brain without any side apparent effects. Mice that previously exhibited signs of Alzheimer’s regained their sense of smell and also showed memory and cognitive improvement. After one year of phage treatment, the treated mice had 80 percent fewer plaques than untreated mice.
Using natural biological agents to treat human illness is an attractive idea that has been used since before recorded history. Bacteriophage therapy, first proposed 90 years ago, could be a viable treatment for the next 90 years..
Interesting Links:
Virus Cleans Up Food Poisoning Bug – The New Scientist April 24, 2003
Phage Therapy May Help Fight Resistant Bacteria – CBS 5 Report with video
The Evilutionary Biologist – Blog by a bacteriophage biologist
Twisted Bacteria – Another site about phages and related microbiology
US Food and Drug Administration – Questions and answers about phage treatment on meat and poultry
August 14th, 2008 at 12:46 pm
natural therapy…
hi!, nice job, i’m sure you are interesting in the 2008 website awards, please contact me for details, i think this is kind of websites we are looking for…